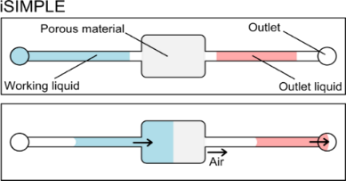
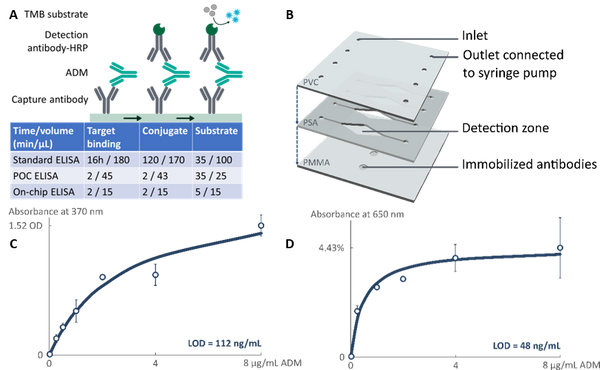
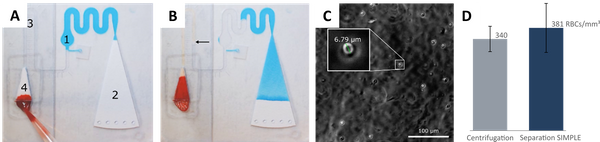
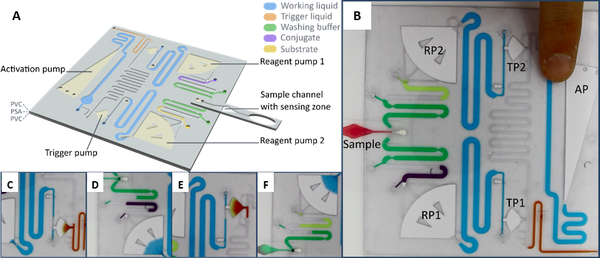
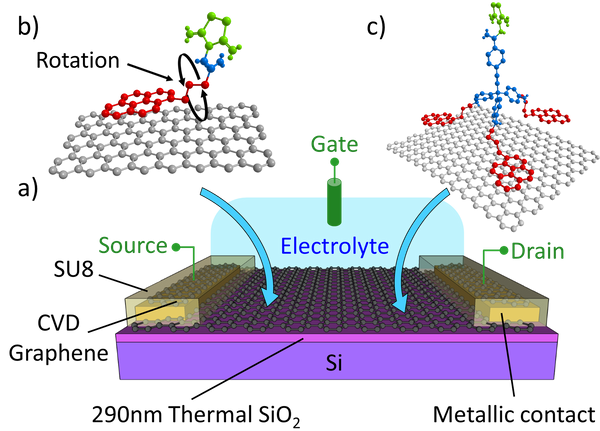
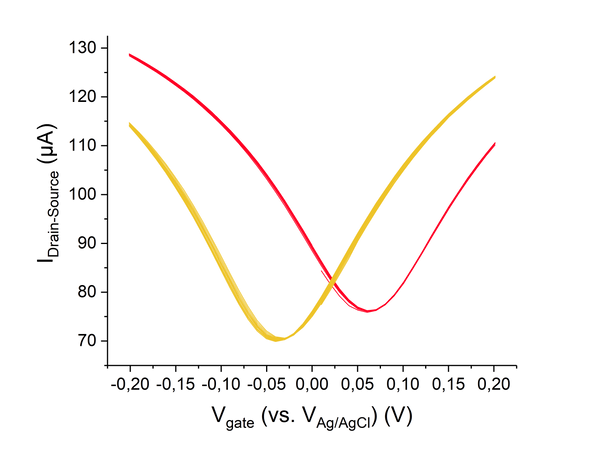
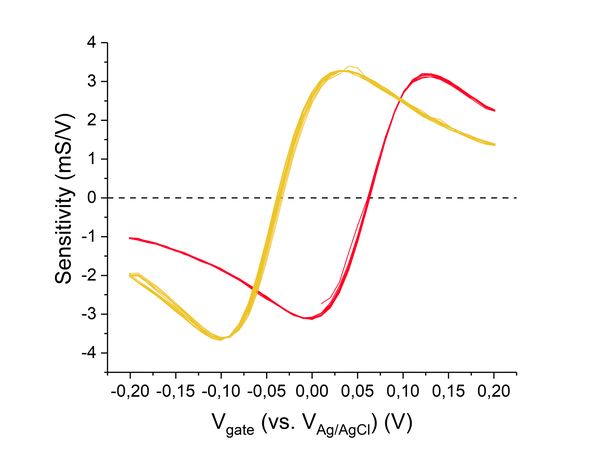
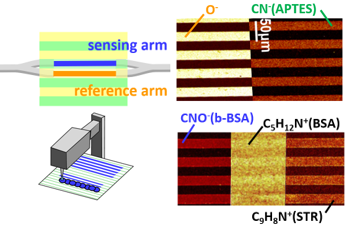
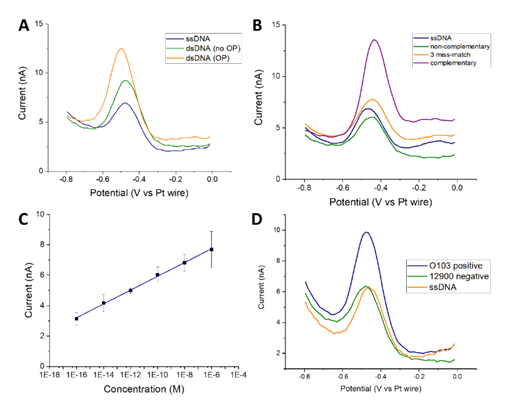
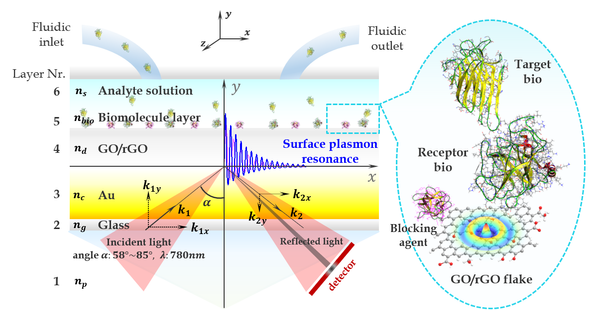
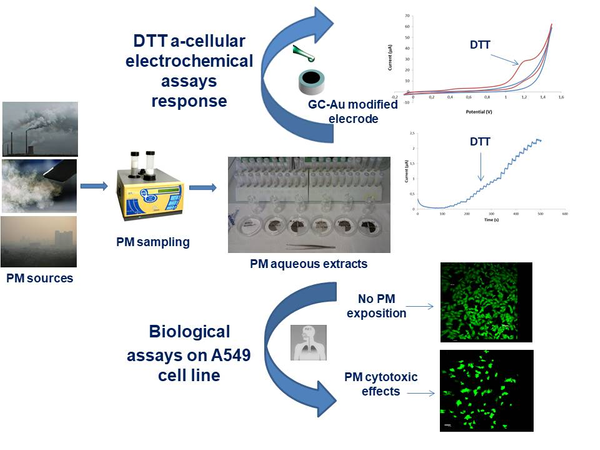
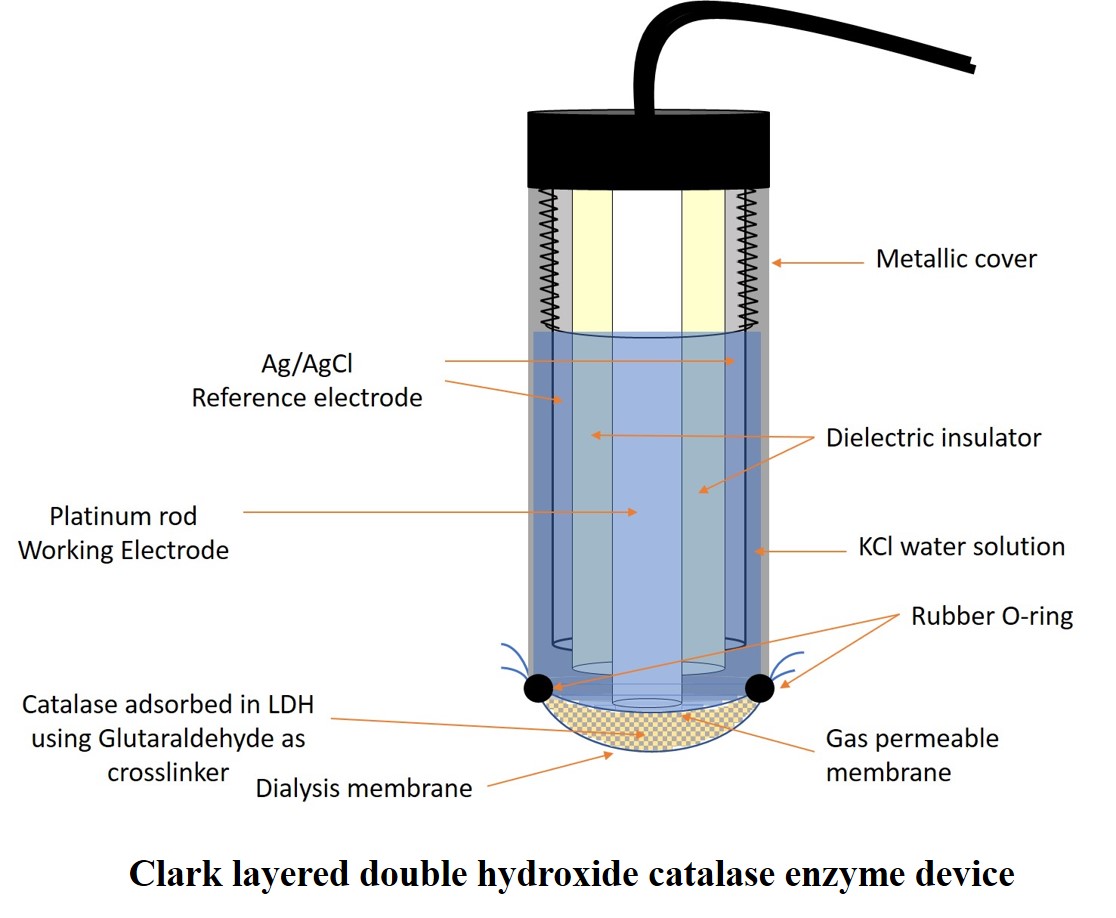
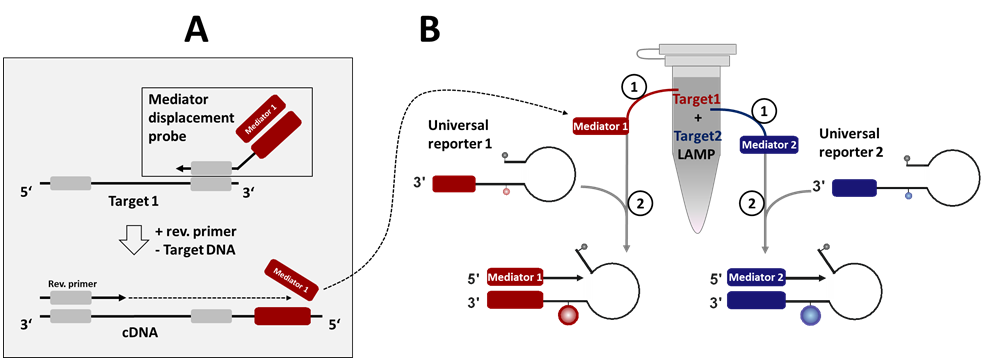
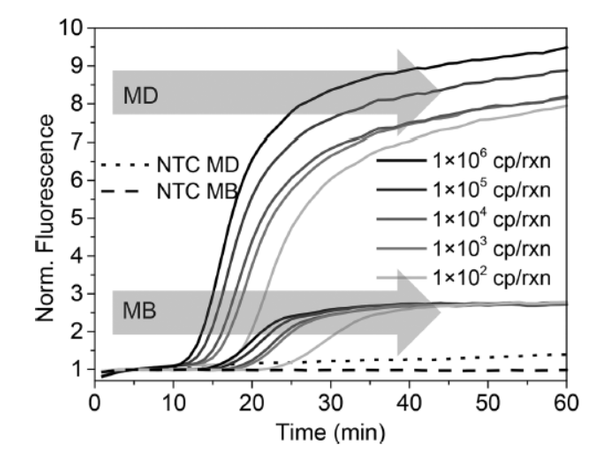
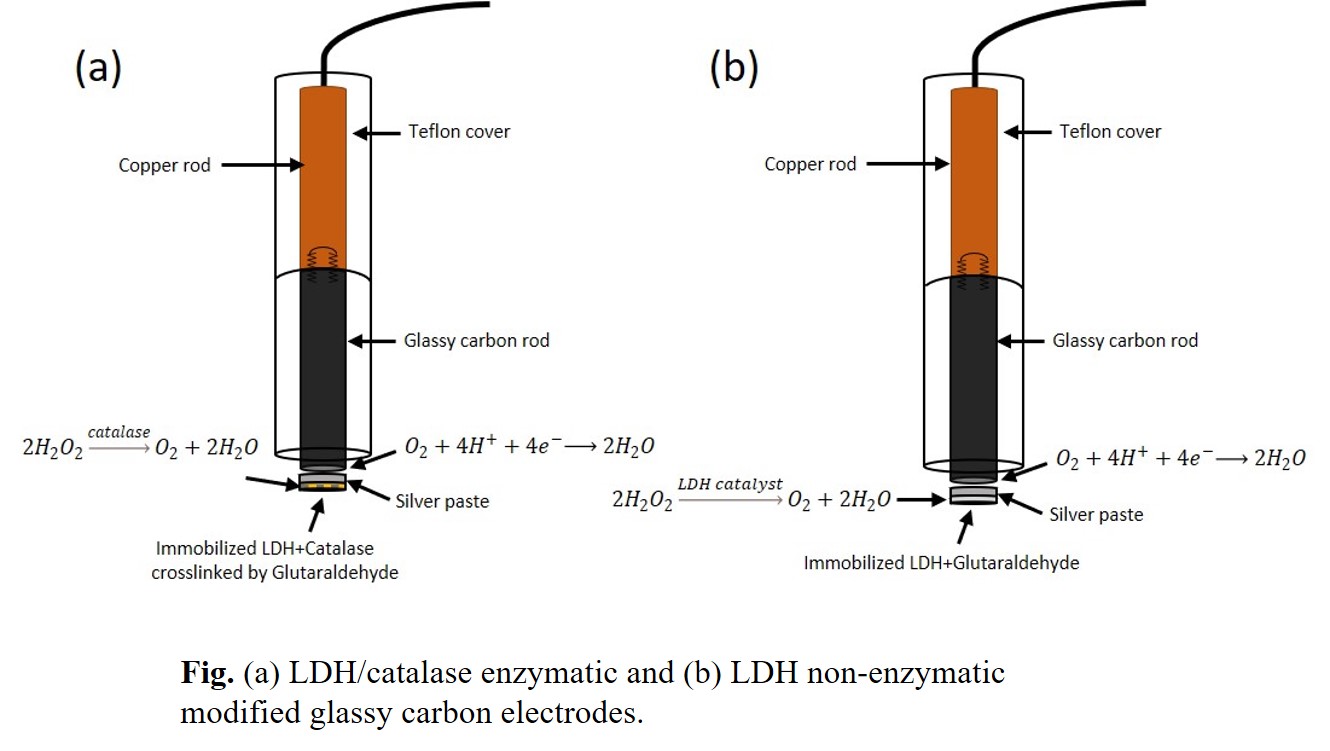
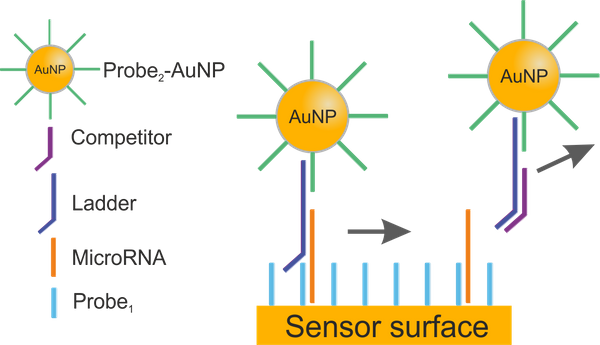
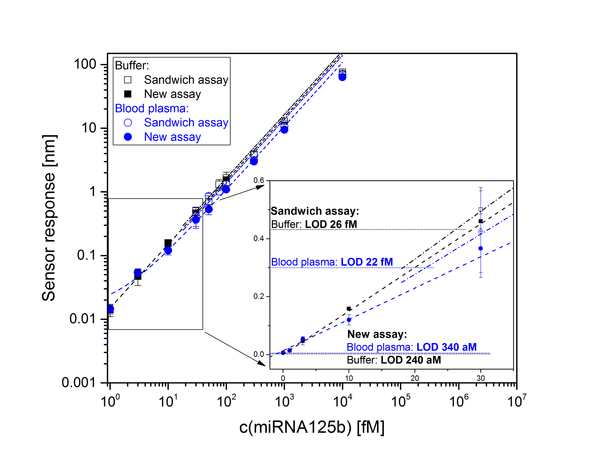
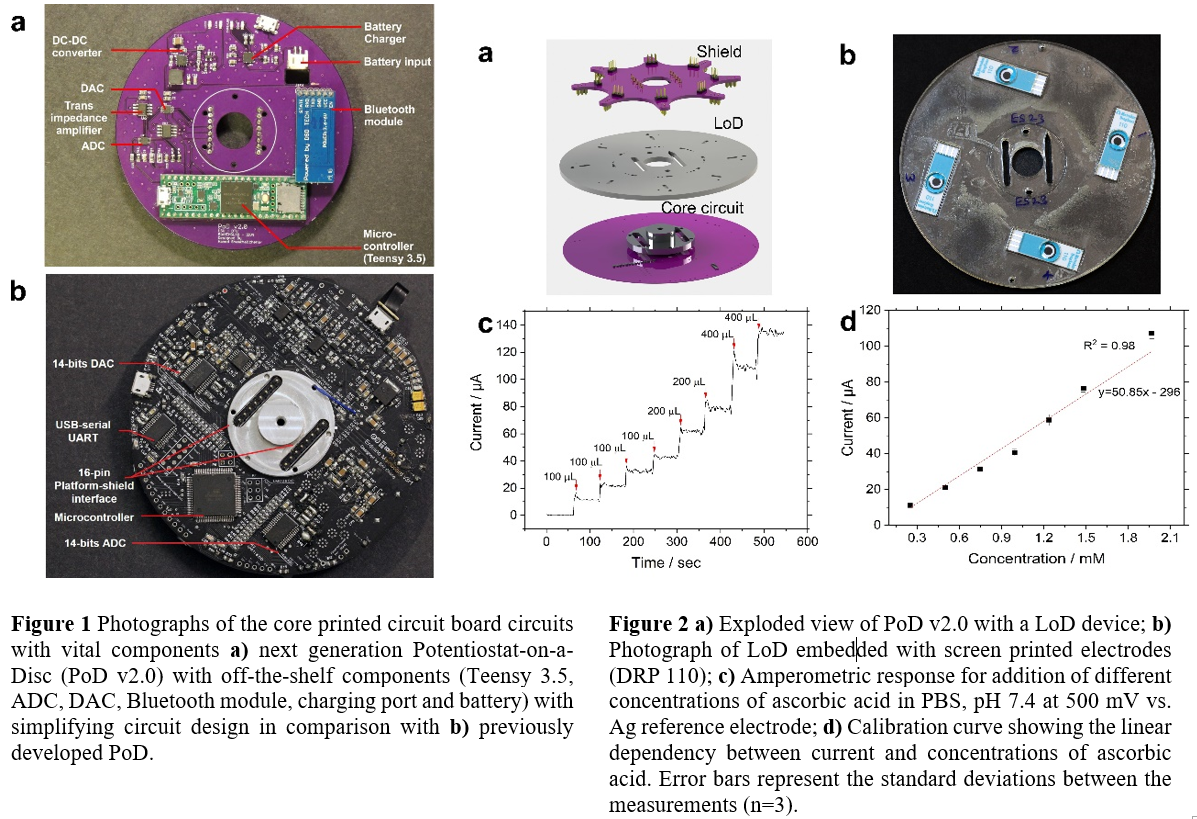
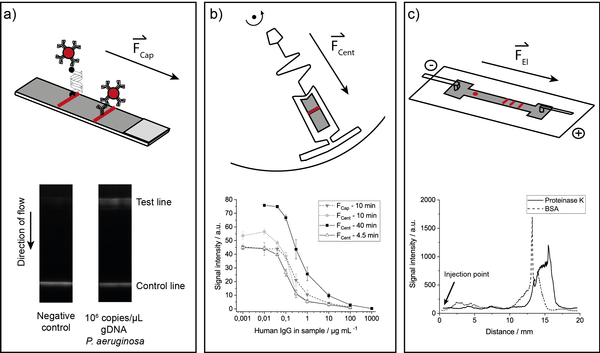
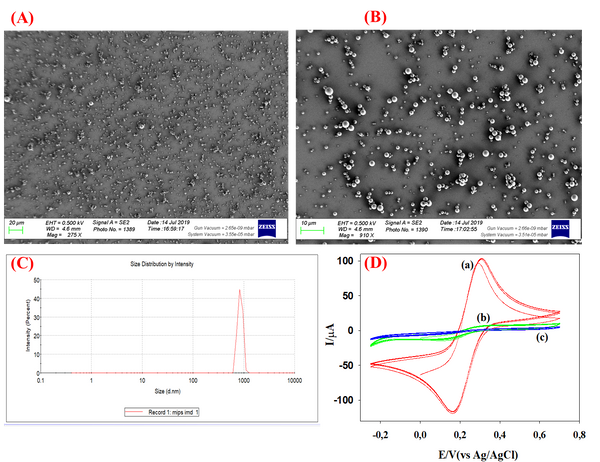
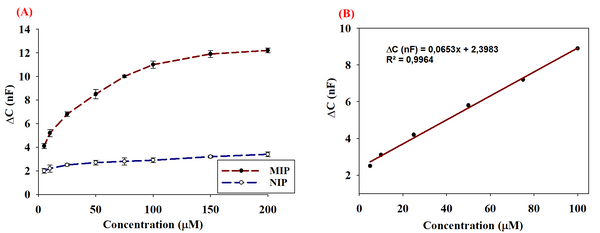
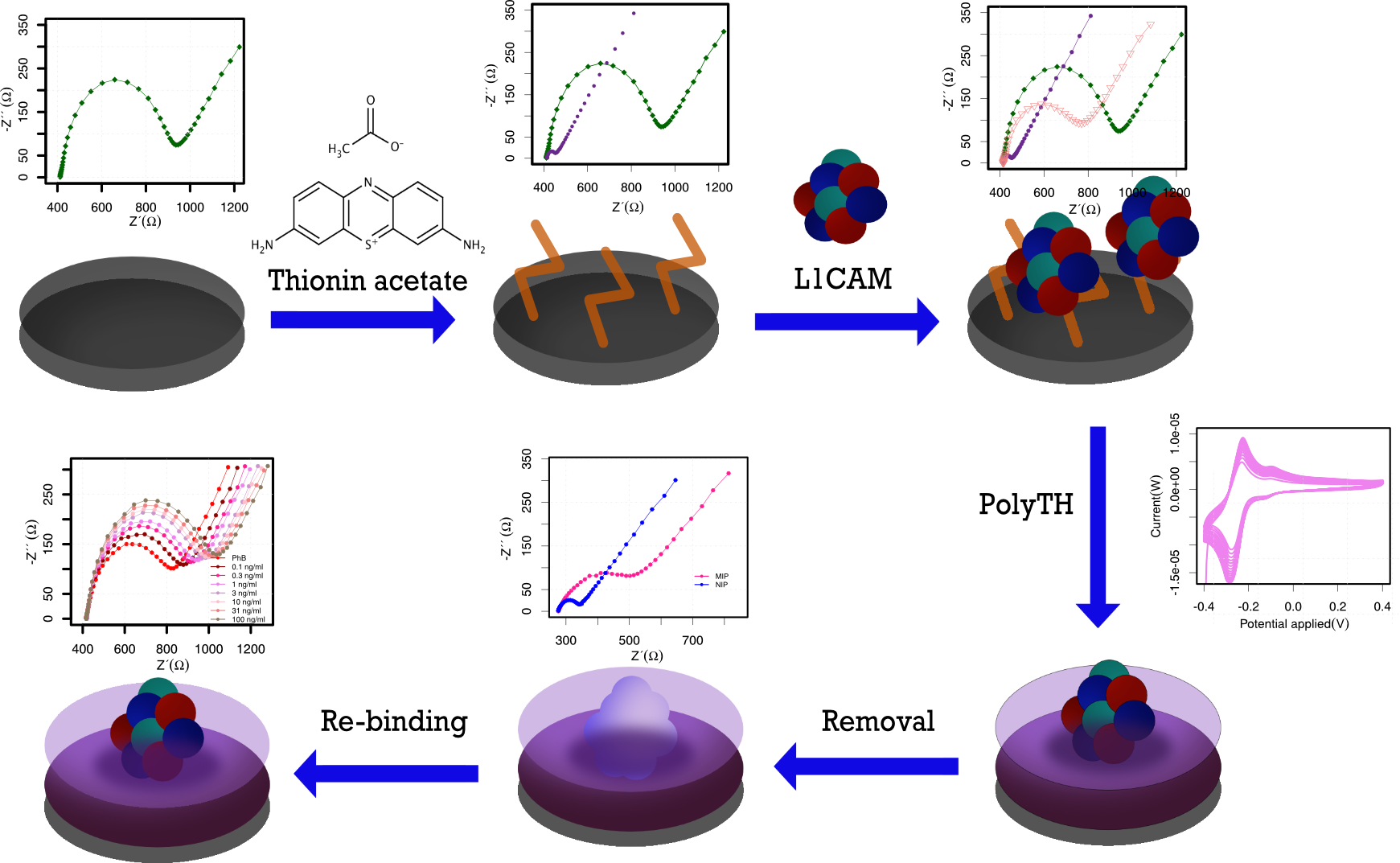
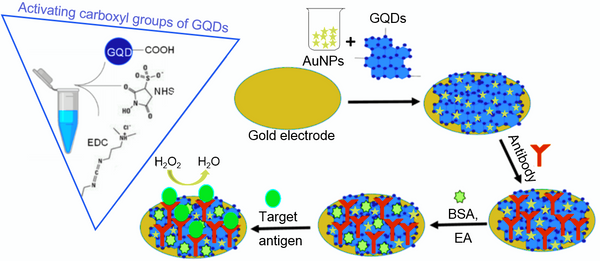
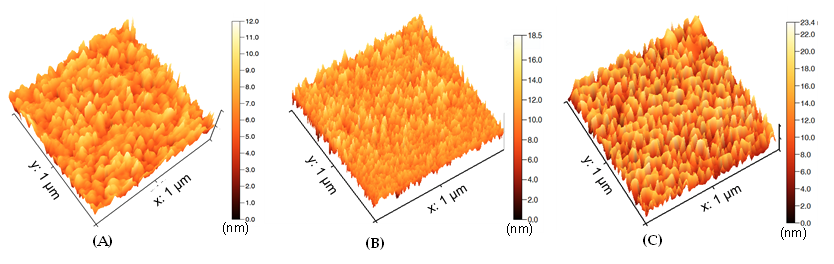
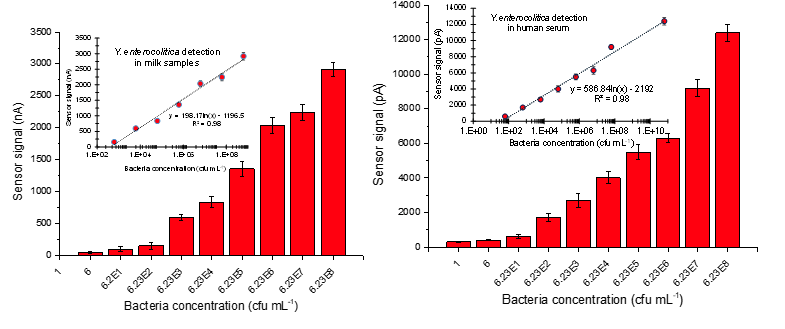
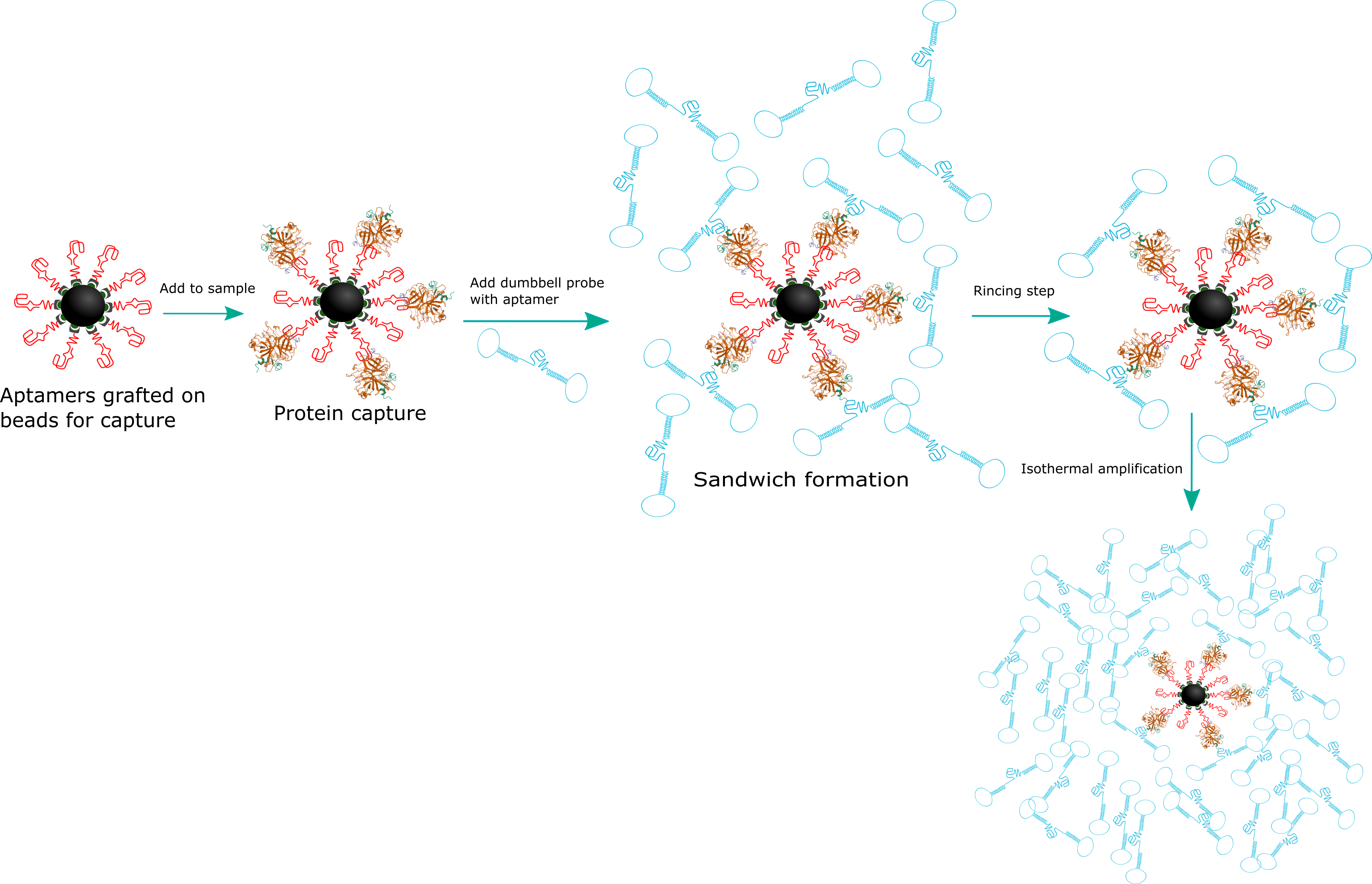
Abstract
Under shear rate conditions prevailing in the microcirculation, vWF- platelet GpIb interaction is a prerequisite of haemostatic plug formation (primary haemostasis: platelet adhesion followed by aggregation). In order to better investigate von Willebrand disease (vWD) and platelet disorders associated with bleeding, we propose to perform real-time evaluation under several shear rates at which the contribution of vWF in platelet plug formation differs.
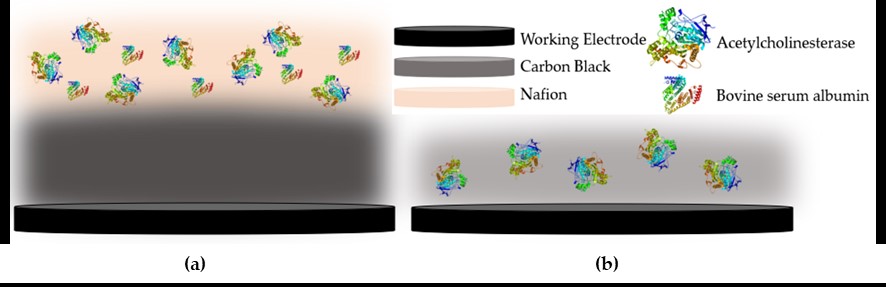
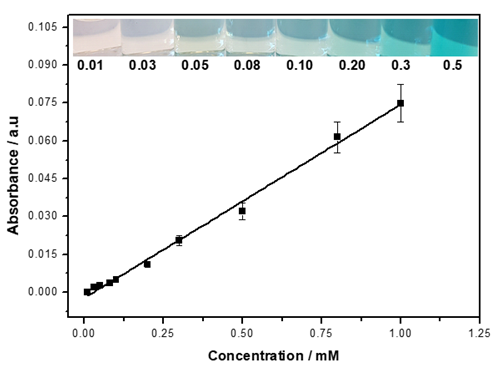
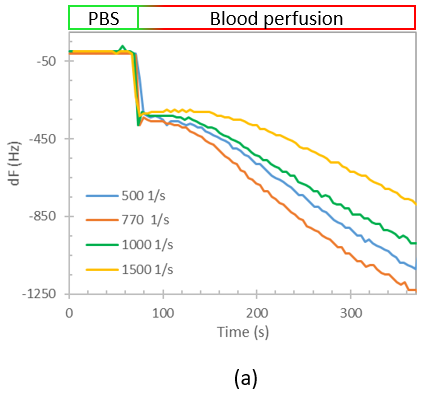
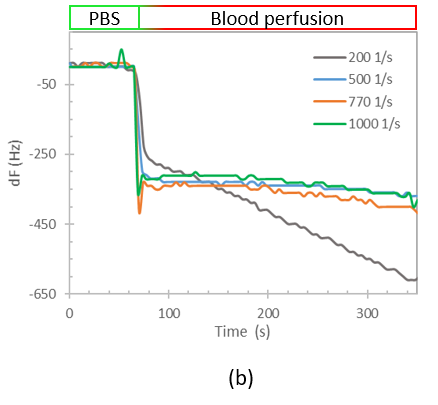
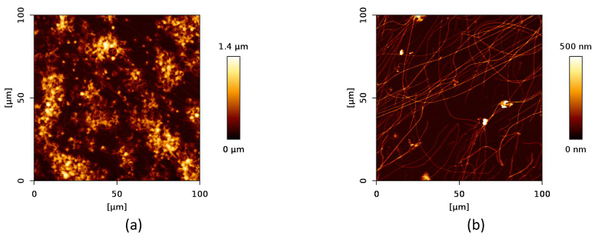
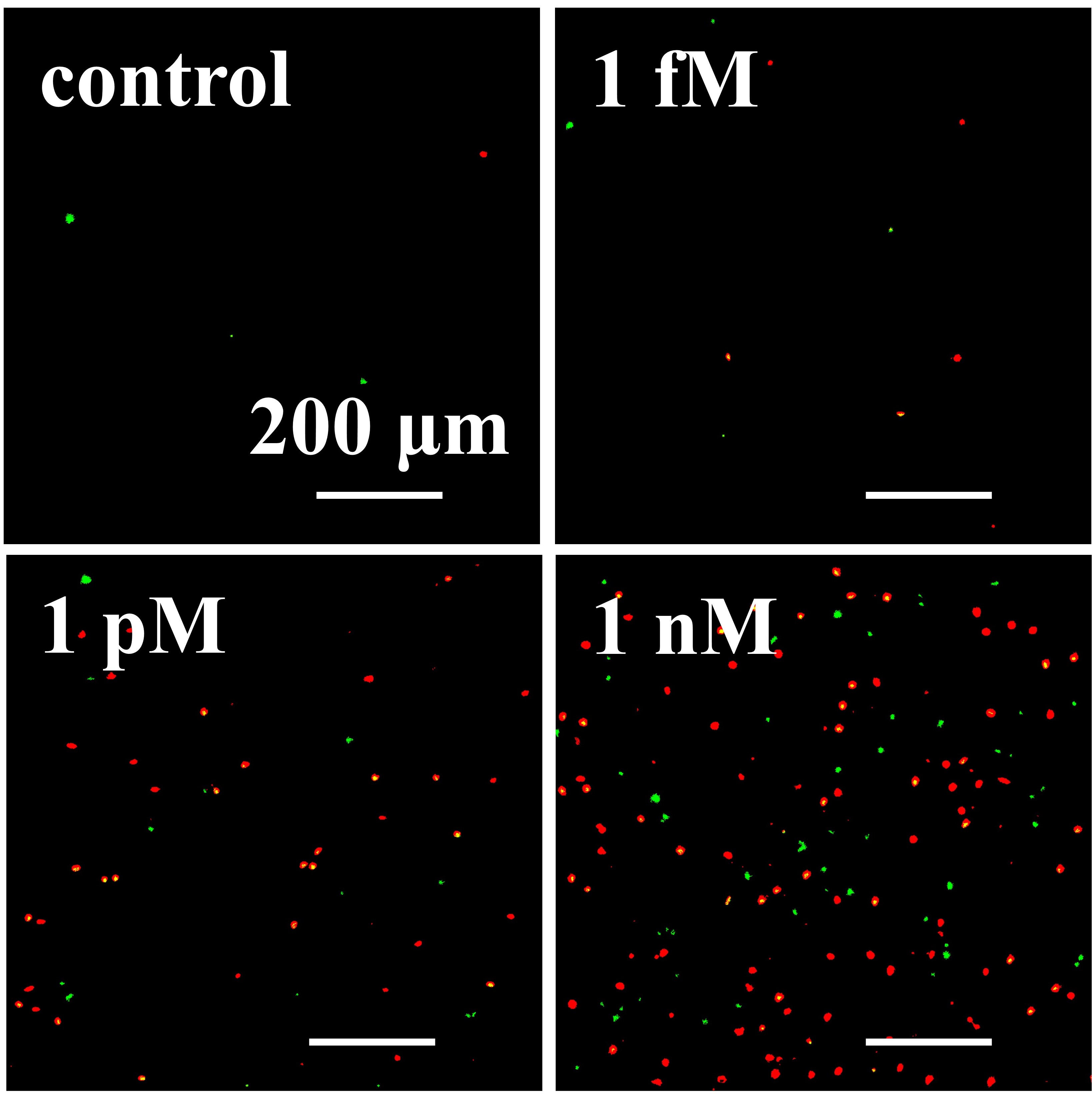
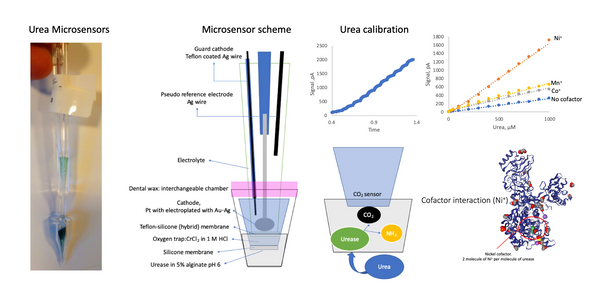
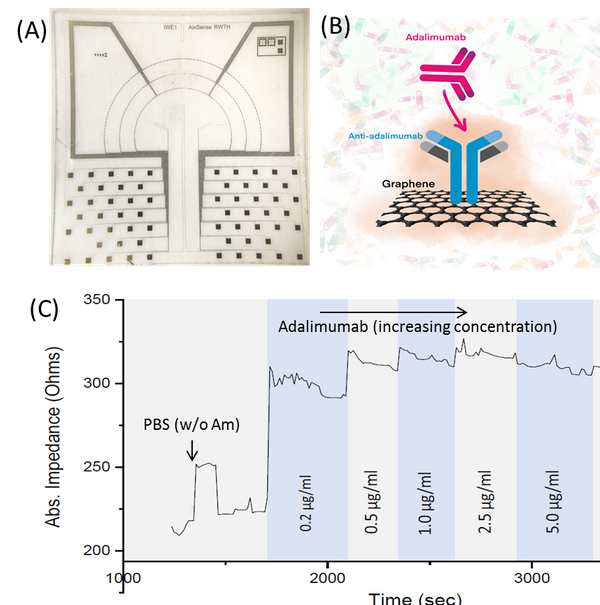
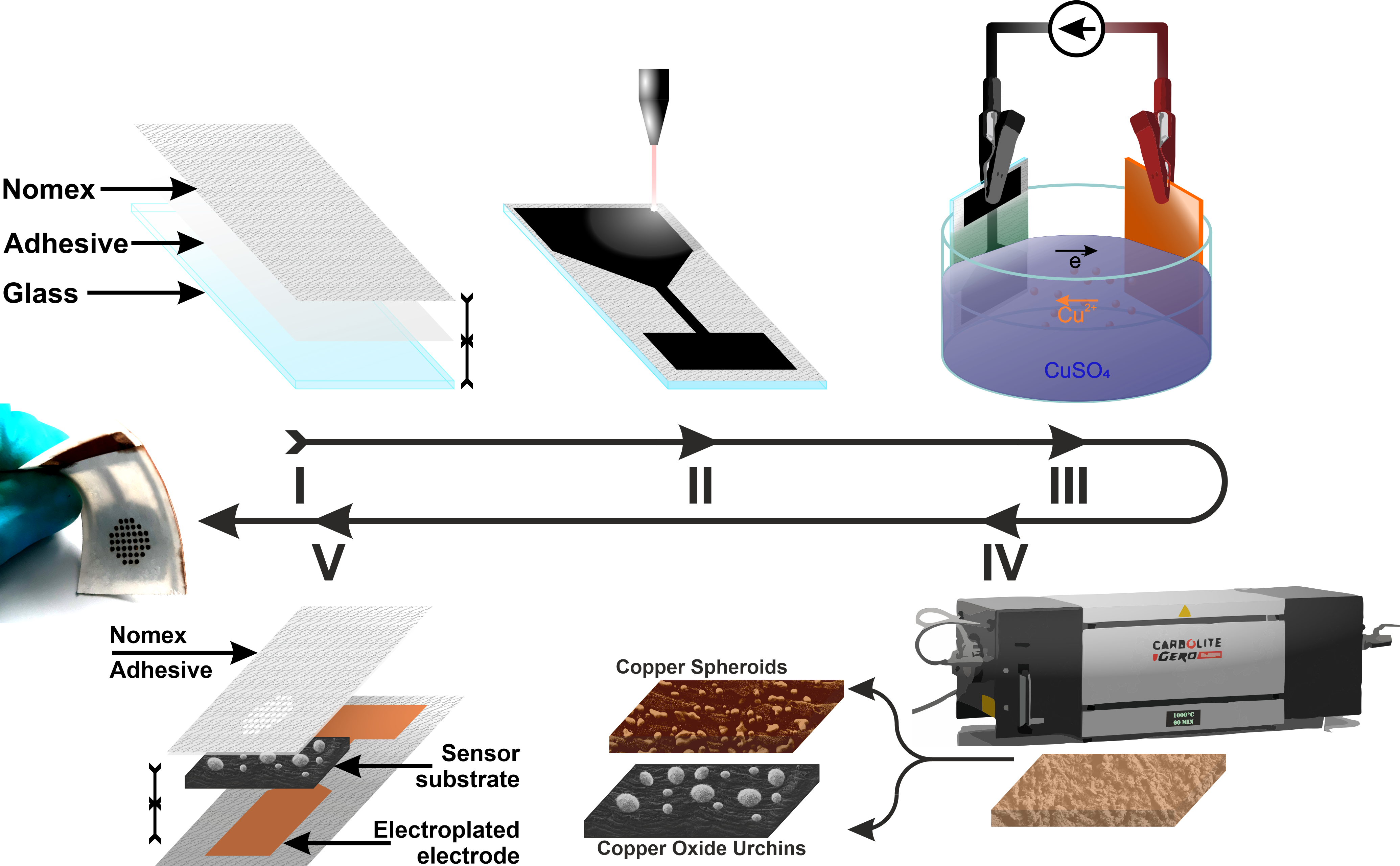
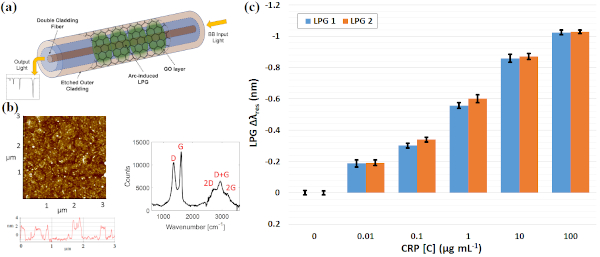
We designed a label-free assessment with a microacoustic biosensor based on quartz crystal microbalance (QCM), Figure 1. The biosensor frequency shift was prior compared with atomic force microscopy (AFM) images of the biointerface after perfusion and found to be in agreement with both coverage and average height of platelet deposits, Figure 2. The test results for 5 minutes perfusion of anticoagulated normal whole blood are shown in Figures 3(a)-4(a). Defective adhesion was induced by inhibition of GpIb platelet receptor with a monoclonal antibody at 10ug/mL. Perfusion results are shown in Figures 3(b)-4(b). Inhibition of GpIb affected initial platelet plug formation in all cases but less at 200 1/s when the contribution of vWF is expected to be the lowest.
The approach we developed is intended to improve evaluation of vWD in a label-free manner by integrative shear dependent phenotyping that is not realised by currently existing devices.
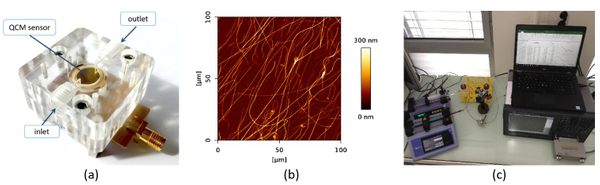
Figure 1. Parallel plate flow perfusion chamber with assembled biosensor (a); AFM image of collagen type I biointerface (b); experimental setup (c).
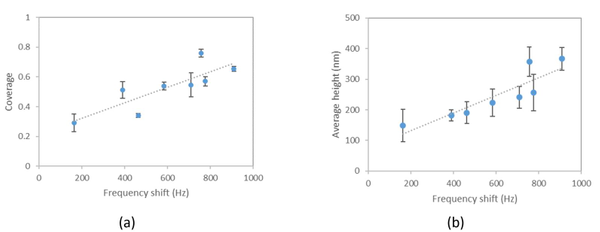
Figure 2. Frequency shift versus AFM measured surface platelet coverage (a) and average height (b) for two blood samples.
Figure 3. Test results of perfusion of whole blood without (a) and with (b) GpIb inhibition.
Figure 4. Representative AFM images of platelet deposits after perfusion (5 minutes) of normal (a) and GpIb inhibited (b) whole blood.

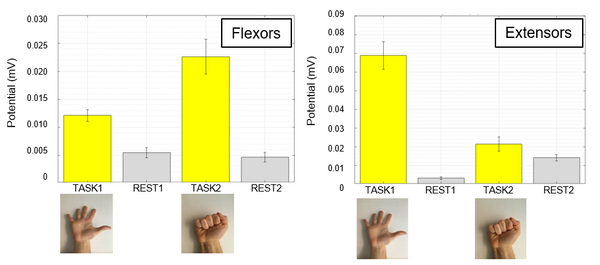
 We validated the acquisition system in a study of sEMG-based fatigue assessment with 6 subjects, well-matched to the current literature [4].
We validated the acquisition system in a study of sEMG-based fatigue assessment with 6 subjects, well-matched to the current literature [4].